Laboratory of RNA Biology and Hepatic Glucose Metabolism
JIVAN (Jiva-Vijnan-Anweshan-Niketan) — Centre for Research in Biological Sciences, RKMVERI
Pushkar Malakar
Ramanujan Fellow and Assistant Professor
Department of Biomedical Science and Technology
School of Biological Sciences, RKMVERI
Ramakrishna Mission Ashrama, Narendrapur, Kolkata 700103
West Bengal, India
Contact:
Email:
Research Interests
A. Long Noncoding RNAs:
Human transcriptome studies indicate that greater than 75% of the human genome is transcribed into thousands of lncRNAs. The biological characterization, roles, and molecular functions of majority lncRNAs remain elusive or unstudied. The first lncRNA identified with an accepted role in cancer is MALAT1. Cancer is a condition driven by various genetic and epigenetic changes that result in the uncontrolled proliferation of cells. Many of these alterations that allow cancer cells to over proliferate, and escape cell survival mechanisms map to signaling pathways (Sever et al., Cold Spring Harb Perspect Med. 2015), genomic instability (Weaver and Cleveland, Curr Opin Cell Biol. 2006), cancer glucose metabolism (Liberti et al., Trends Biochem Sci. 2016) and aberrant alternative splicing (Anczukow et al. RNA.2016).
The field of lncRNAs is still new, and we are far from incorporating it into the clinic. The functions of lncRNAs in pathological conditions and in normal physiological conditions are far from fully understood. Despite the rapid increase in the number of lncRNAs, one of the biggest challenges is identifying the sequence and structural elements necessary for long noncoding RNA molecules to execute their cellular functions (Fang et al., Genomics, Proteomics & Bioinformatics, 2016).
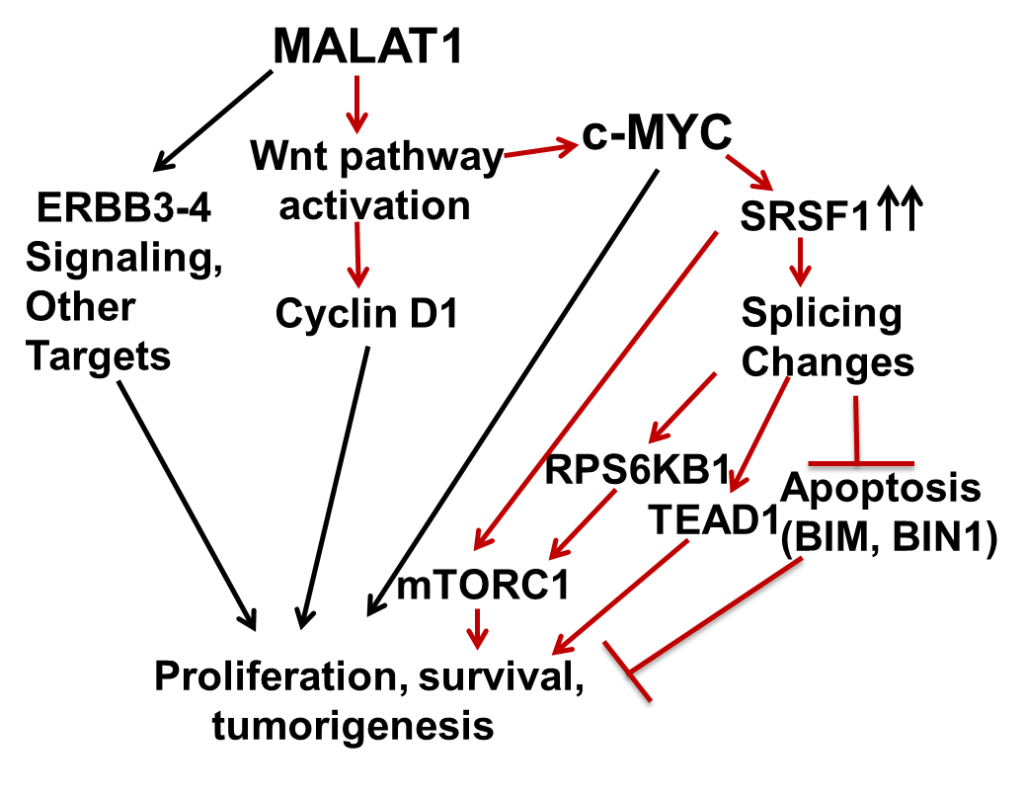
During my postdoctoral training we showed that Long Noncoding RNA MALAT1 Promotes Hepatocellular Carcinoma Development by SRSF1 Upregulation and mTOR Activation (Cancer Res. 2017 Mar 1; 77(5):1155-1167). The summary of the work is mentioned below.
B. Hepatic Glucose Metabolism: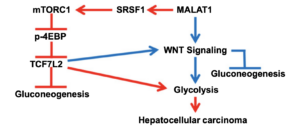
Altered glucose metabolism is one of the first identified hallmarks of cancer, discovered by Otto Warburg in the late 1920s (Cell 2011;144:646–74.) Cancer glucose metabolism an integral part of cancer research. Cancer cells predominantly carry out glycolysis in the cytosol rather than oxidative phosphorylation through the TCA cycle in the mitochondria (Mol Cell 2015; 59:491–501.) It is generally believed that in most cancers, oncogenic lesions are largely the cause of enhanced glycolysis and the “Warburg effect”. Gluconeogenesis is essentially the reverse of the glycolysis pathway and usually occurs in the liver when blood glucose levels drop and the liver regenerates glucose, sending it to other tissues (Compr Physiol 2014; 4:177–97.). The field of glucose metabolism is still new, and we are far from incorporating it into the clinic. The functions of hepatic glucose metabolism in pathological conditions and in normal physiological conditions are far from fully understood.
In one of the projects, we studied the role of MALAT1 in regulating cancer glucose metabolism. MALAT1 upregulation in HCC resulted in increased expression of glycolytic genes and downregulation of gluconeogenic enzymes. In Hepatocellular carcinoma (HCC) the regulation of cancer glucose metabolism by MALAT1 is dependent on the metabolic transcription factor TCF7L2. MALAT1 upregulates the protein levels of TCF7L2 by increased mTOR mediated translation (Cancer Res. 2019 May 15;79(10):2480-2493.). The summary of the work is mentioned below.
C. Alternative Splicing: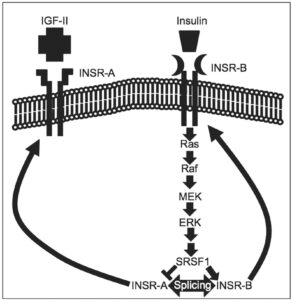
Alternative splicing is a key process that produces different proteins from the same gene. There is a clear correlation between many diseases and the alterations and mistakes in splicing regulation produced by splicing factors. Splicing Factors are trans acting RNA Binding Proteins. Very few RNA Binding Proteins (RBPs) are associated with altered glucose metabolism or diabetic complications. In addition, their precise contributions to impaired glucose metabolism are still emerging (Fredericks et al. 2015). The field of alternative splicing is still new and expect few alternative splicing targets we are far from incorporating it into the clinic. The functions of alternative splicing in pathological conditions and in normal physiological conditions are far from fully understood. One of the biggest challenges in the field of alternative splicing is to identify the regulators of alternative splicing to execute their cellular functions.
In one of my projects, we showed that insulin induces INSR exon 11 inclusion (INSR-B) in pancreatic beta cells through activation of the Ras-MAPK/ERK signaling pathway and up-regulation of the splicing factor SRSF1. Induction of exon 11 skipping (INSR-A) inhibited the MAPK/ERK signaling pathway, sensitizing the pancreatic β-cell line MIN6 to stress-induced apoptosis and lipotoxicity (Sci Rep. 2016 Aug 16; 6:31222). The summary of the work is mentioned below.
Ongoing projects
Role of Long Noncoding RNAs in normal physiological and pathological conditions.
Recent Publications
- Long noncoding RNA, PURPL is associated with aneuploidy and its magnitude of expression level is dependent on P53 status. Malakar P ##. Front Cell Dev Biol. 2024 Dec 23; 12:1410308. # #As Single and Corresponding Author.
- Unleashing innovation: 3D-printed biomaterials in bone tissue engineering for repairing femur and tibial defects in animal models – a systematic review and meta-analysis. Sagar N, Chakravarti B, Maurya SS, Nigam A, Malakar P, Kashyap R. Front Bioeng Biotechnol. 2024 Sep 23; 12:1385365.
- Microplastics in the soil-water-food nexus: Inclusive insight into global research findings. Garai S, Bhattacharjee C, Sarkar S, Moulick D, Dey S, Jana S, Dhar A, Roy A, Mondal K, Mondal M, Mukherjee S, Ghosh S, Singh P, Ramteke P, Manna D, Hazra S, Malakar P, Banerjee H, Brahmachari K, Hossain A. Sci Total Environ. 2024 Oct 10; 946:173891.
- Characterization of polyploidy in cancer: Current status and future perspectives. Ghosh S, Choudhury D, Ghosh D, Mondal M, Singha D, Malakar P *. Int J Biol Macromol. 2024 May;268(Pt 1):131706. * As Corresponding Author.
- Polyploidy and mTOR signaling: a possible molecular link. Choudhury D, Ghosh D, Mondal M, Singha D, Pothuraju R, Malakar P *. Cell Commun Signal. 2024 Mar 27;22(1):196. * As Corresponding Author.
- Gum acacia dietary fiber: Significance in immunomodulation, inflammatory diseases, and cancer. Barkeer S, Pothuraju R, Malakar P, Pimentel TC, Siddiqui JA, Nair SA. Phytother Res. 2024 Mar;38(3):1509-1521.
- The nexus of long noncoding RNAs, splicing factors, alternative splicing and their modulations. Malakar P#, Shukla S, Mondal M, Kar RK, Siddiqui JA. RNA Biol. 2024 Jan;21(1):1-20. # As first and corresponding author
- The role of alternative splicing and splicing factors in diabetes: Current status and future perspectives. Singha D, Mondal M, Ghosh D, Choudhury D, Chakravarti B, Kar RK, Malakar P*. Wiley Interdiscip Rev RNA. 2024;15(1): e1831. *As Corresponding Author.
- Pan-cancer integrated bioinformatic analysis of RNA polymerase subunits reveal RNA Pol I member CD3EAP regulates cell growth by modulating autophagy. Bhandari N, Acharya D, Chatterjee A, Mandve L, Kumar P, Pratap S, Malakar P, Shukla SK. Cell Cycle. 2023 Oct 5:1-17.
- Giant cells: multiple cells unite to survive. Hazra S, Kalyan Dinda S, Kumar Mondal N, Hossain SR, Datta P, Yasmin Mondal A, Malakar P, Manna D. Front Cell Infect Microbiol. 2023 Sep 5; 13:1220589.
- Glutamine regulates the cellular proliferation and cell cycle progression by modulating the mTOR mediated protein levels of β-TrCP. Malakar P#, Singha D, Choudhury D, Shukla S. Cell Cycle. 2023 Sep 28:1-14. # As First and Corresponding Author
- A Possible Novel Therapeutic Targets of Selinexor in Acute Lymphoblastic Leukemia: A Comprehensive Review. Malakar P #, Sagar N, Chakravarti B, Mondal M, Singha D, Kar R. J Clin Transl Res. 2023;9(6):423-432. # As First and Corresponding Author
- Molecular signaling network and therapeutic developments in breast cancer brain metastasis. Benjamin M, Malakar P, Sinha RA, Nasser MW, Batra SK, Siddiqui JA, Chakravarti B. Adv Cancer Biol Metastasis. 2023 Jul; 7:100079
- Emerging frontiers of antibiotics use and their impacts on the human gut microbiome. Kumari R, Yadav Y, Misra R, Das U, Das Adhikari U, Malakar P, Dubey GP. Microbiol Res. 2022 Oct; 263:127127. (7 Citations).
- Malakar P, Stein I, Saragovi A, Winkler R, Stern-Ginossar N, Berger M, Pikarsky E, Karni R (2019). Long Noncoding RNA MALAT1 regulates cancer glucose metabolism by enhancing mTOR-mediated TCF7L2 translation. Cancer Res. 79(10):2480-2493. (120 Citations).
- Malakar P, Shilo A, Mogilevsky A, Stein I, Pikarsky E, Nevo Y, Benyamini H, Elgavish S, Zong X, Prasanth KV, Karni R (2017). Long Noncoding RNA MALAT1 Promotes Hepatocellular Carcinoma Development by SRSF1 Upregulation and mTOR Activation. Cancer Res. 77(5):1155-1167. (227 Citations).
- Malakar P, Chartarifsky L, Hija A, Leibowitz G, Glaser B, Dor Y, Karni R (2016). Insulin receptor alternative splicing is regulated by insulin signaling and modulates beta cell survival. Sci Rep. Aug 16; 6: 31222. (58 Citations).

